 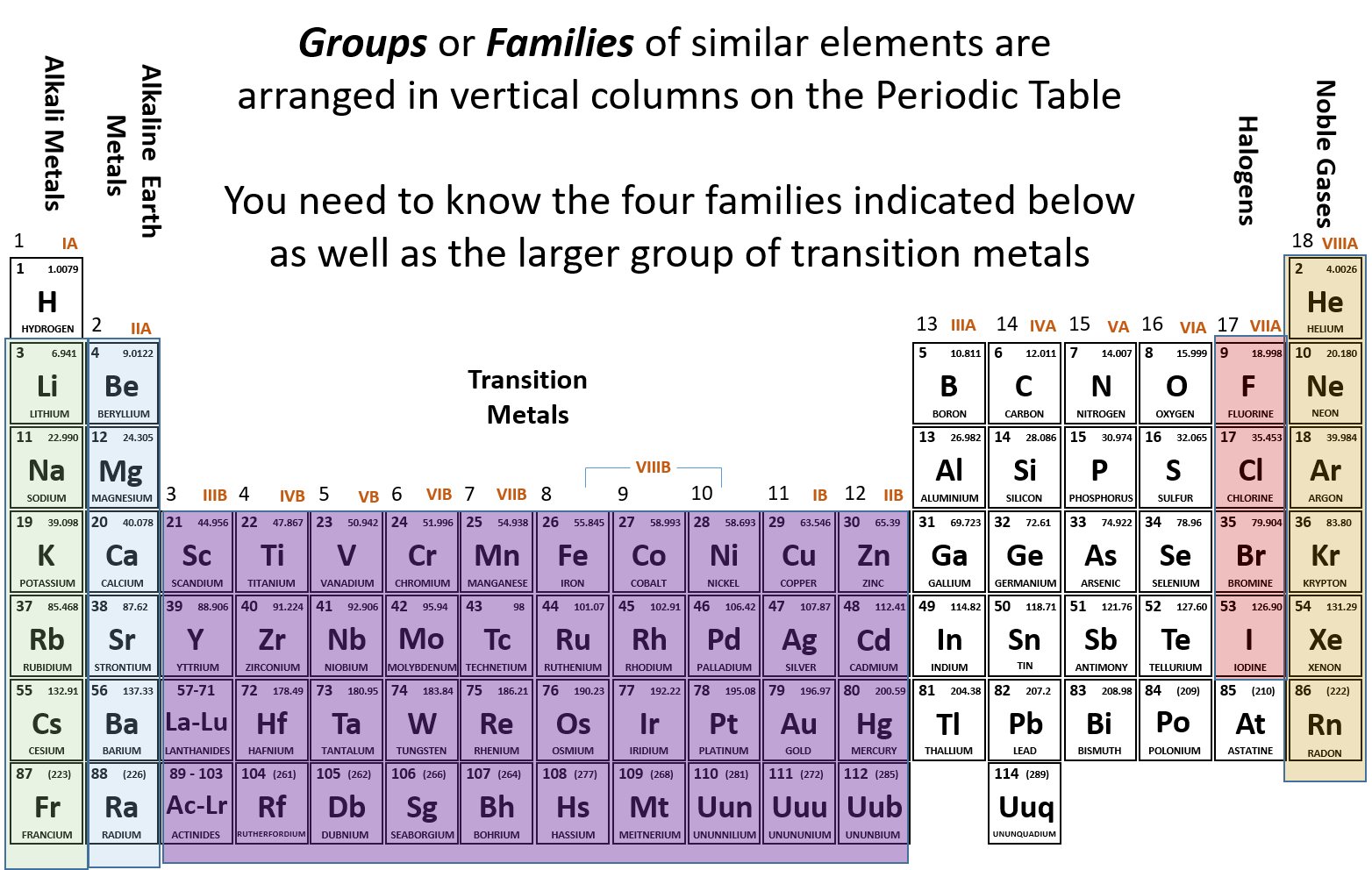
Sodium chloride, ordinary table salt, is the result of a chemical reaction between the metal sodium and the halogen chlorine. Electron donors such as metals like to combine with halogens to make stable compounds. contains non-metal close non-metal Element that is a poor conductor. Halogens tend to form stable arrangements called diatomic molecules in which two of the same halogen element share an electron. Group 7 close group 7 The vertical column of non-metal elements next to group 0 in the periodic table, also called the halogens. Halogens, because they need one more electron to have a stable arrangement, generally try to grab electrons from other elements therefore, halogens are considered electronegative elements. All other elements need to find situations in which they can either donate or receive electrons to achieve their full complement. Exception: Hydrogen is a nonmetal which is located on the left top corner of the Periodic table. In nature, the noble gases generally do not form compounds with any other element. Where are the most reactive nonmetal elements found on the periodic table A. Augby Jay Nonmetals are located on the upper right side of the Periodic table (see above image). Its oxidation state is always -1 except in its elemental, diatomic state (in which its oxidation state is zero). Fluorine also has a relatively small atomic radius. It appears as a pale yellow gas at room temperature. 
For example, the noble gases, which contain eight electrons in the outermost energy level, are extremely stable elements because they already have their full complement of electrons. Fluorine is the most electronegative element in the periodic table. The tendency of atoms is to fill this outermost shell to capacity to attain a stable arrangement of electrons. These valence electrons are the cause of an element’s reactivity. The further down the group you go, the less reactive the element is. most reactive metals and non-metals respectively how the structures of all. The most reactive element from group seven is fluorine which is at the top of that section of the periodic table. Periodic Table lists the elements of the Universe in order of increasing. Halogens have seven valence electrons, which means that they have seven electrons in their outermost shells. The halogens include fluorine, chlorine, bromine, iodine and astatine. The elements fluorine, chlorine, bromine, iodine and astatine make up the halogen family.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
